SUMMARY: Prostate Cancer is the most common cancer in American men and approximately 233,000 new cases will be diagnosed in 2014 and close to 30,000 men will die of the disease. The primary systemic intervention for patients with advanced prostate cancer is Androgen Deprivation Therapy (ADT). This can be accomplished by either surgical castration (bilateral orchicetomy) or medical castration, using LHRH (GnRH- Gonadotropin-Releasing Hormone) agonists. Majority of these patients will eventually develop progressive disease (Castrate Resistant Prostate cancer – CRPC), due to enhanced autocrine and /or paracrine synthesis of androgens or androgen precursors in the tumor micro environment. This has lead to the development of novel compounds that decrease androgen synthesis as well as androgen signaling in patients with CRPC.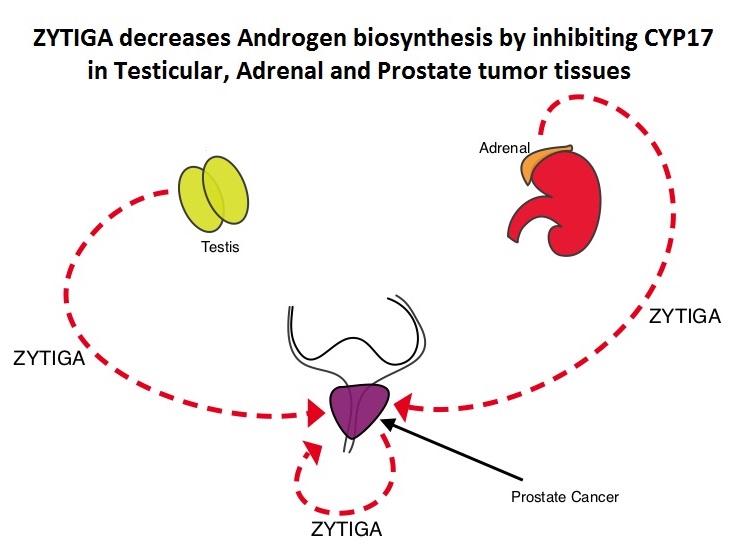 Abiraterone acetate (ZYTIGA®) is a novel, targeted, oral androgen biosynthesis inhibitor that decreases androgen production in the adrenal glands, testes and prostate cancer cells by inhibiting a steroidal enzyme CYP17A. Phase III trials have demonstrated that treatment with ZYTIGA® prolongs overall survival in metastatic CRPC patients, who had progressed after TAXOTERE® (Docetaxel) therapy, as well as those who are chemotherapy naive. ZYTIGA® delays deterioration of performance status, progression of fatigue and pain as well as development of skeletal related events, in TAXOTERE® refractory patients. It is important that any treatment considered for patients with asymptomatic or mildly symptomatic CRPC improves overall survival without negatively impacting Quality of Life. To address this further, the authors analyzed patient reported data related to pain and Quality of Life from a large randomized clinical trial. Of the 1088 chemotherapy-naïve, asymptomatic or mildly symptomatic CRPC patients randomized in this double-blind study, 546 patients received ZYTIGA® 1000 mg PO plus prednisone 5 mg twice daily and 542 patients received placebo plus prednisone. At the time of the planned interim analysis, ZYTIGA® improved radiographic progression-free survival, overall survival, and significantly delayed the initiation of chemotherapy. The authors in this publication reported the data related to pain and Quality of Life of these patients, at the time of the second preplanned interim analysis. Pain was assessed with the Brief Pain Inventory-Short Form (BPI-SF) questionnaire, which is a validated instrument to assess pain and Health Related Quality of Life (HRQoL) was measured with the Functional Assessment of Cancer Therapy—Prostate (FACT-P) questionnaire, which is a validated tool for metastatic CRPC. At a median follow-up of 22.2 months, the median time to progression of pain intensity was longer in patients receiving ZYTIGA® plus prednisone vs placebo plus prednisone (26.7 months vs 18.4 months, HR=0.82, P=0.049). The median time for pain to progress and interfere with daily activities was 10.3 months for ZYTIGA® vs 7.4 months for placebo (HR= 0.79, P=0.005). The median time to deterioration of HRQoL was longer in patients receiving ZYTIGA® plus prednisone vs those receiving placebo plus prednisone, as assessed by the FACT-P total score (12.7 months vs 8.3 months, HR=0.78, P=0.003). The authors concluded that ZYTIGA® given along with prednisone delays patient-reported pain progression and deterioration of HRQol, in chemotherapy-naive patients with metastatic CRPC, without compromising efficacy. Basch E, Autio K, Ryan CJ, et al. The Lancet Oncology 2013;14:1193 -1199
Abiraterone acetate (ZYTIGA®) is a novel, targeted, oral androgen biosynthesis inhibitor that decreases androgen production in the adrenal glands, testes and prostate cancer cells by inhibiting a steroidal enzyme CYP17A. Phase III trials have demonstrated that treatment with ZYTIGA® prolongs overall survival in metastatic CRPC patients, who had progressed after TAXOTERE® (Docetaxel) therapy, as well as those who are chemotherapy naive. ZYTIGA® delays deterioration of performance status, progression of fatigue and pain as well as development of skeletal related events, in TAXOTERE® refractory patients. It is important that any treatment considered for patients with asymptomatic or mildly symptomatic CRPC improves overall survival without negatively impacting Quality of Life. To address this further, the authors analyzed patient reported data related to pain and Quality of Life from a large randomized clinical trial. Of the 1088 chemotherapy-naïve, asymptomatic or mildly symptomatic CRPC patients randomized in this double-blind study, 546 patients received ZYTIGA® 1000 mg PO plus prednisone 5 mg twice daily and 542 patients received placebo plus prednisone. At the time of the planned interim analysis, ZYTIGA® improved radiographic progression-free survival, overall survival, and significantly delayed the initiation of chemotherapy. The authors in this publication reported the data related to pain and Quality of Life of these patients, at the time of the second preplanned interim analysis. Pain was assessed with the Brief Pain Inventory-Short Form (BPI-SF) questionnaire, which is a validated instrument to assess pain and Health Related Quality of Life (HRQoL) was measured with the Functional Assessment of Cancer Therapy—Prostate (FACT-P) questionnaire, which is a validated tool for metastatic CRPC. At a median follow-up of 22.2 months, the median time to progression of pain intensity was longer in patients receiving ZYTIGA® plus prednisone vs placebo plus prednisone (26.7 months vs 18.4 months, HR=0.82, P=0.049). The median time for pain to progress and interfere with daily activities was 10.3 months for ZYTIGA® vs 7.4 months for placebo (HR= 0.79, P=0.005). The median time to deterioration of HRQoL was longer in patients receiving ZYTIGA® plus prednisone vs those receiving placebo plus prednisone, as assessed by the FACT-P total score (12.7 months vs 8.3 months, HR=0.78, P=0.003). The authors concluded that ZYTIGA® given along with prednisone delays patient-reported pain progression and deterioration of HRQol, in chemotherapy-naive patients with metastatic CRPC, without compromising efficacy. Basch E, Autio K, Ryan CJ, et al. The Lancet Oncology 2013;14:1193 -1199

